Our Research
The brain must transform sensory signals into a neural code that drives perception and behavior.
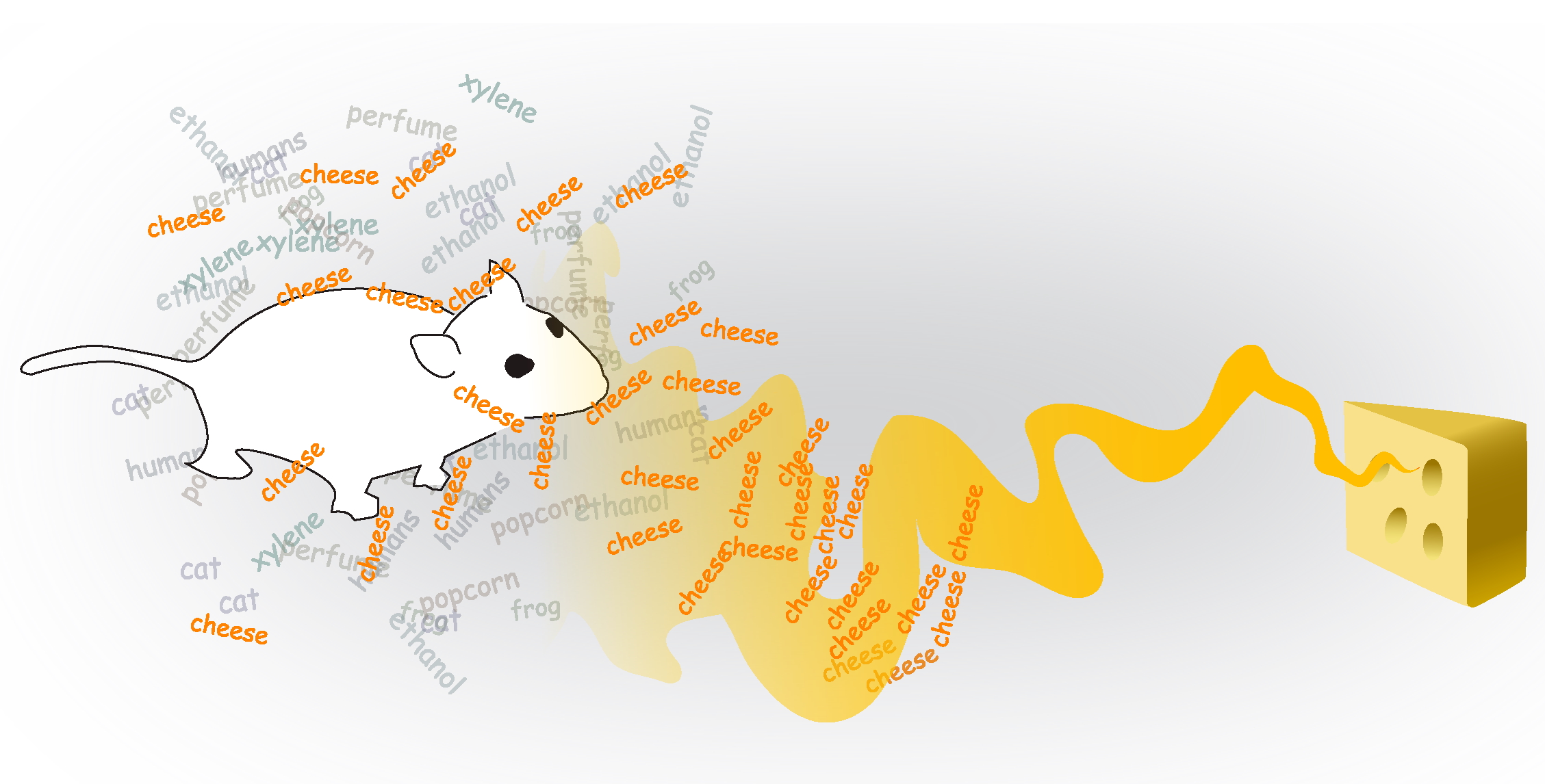
Background Segmentation
Distinguishing a percept from others in the background.
Concentration Invariance
Recognizing an odor as the same whether it's near or far away.
Concentration Sensitivity
Optimizing detection of the concentration changes.
Our hypothesis is that the olfactory bulb is critically involved in carrying out these functions.
Odor detection begins by the transmission of sensory cues into the olfactory bulb.
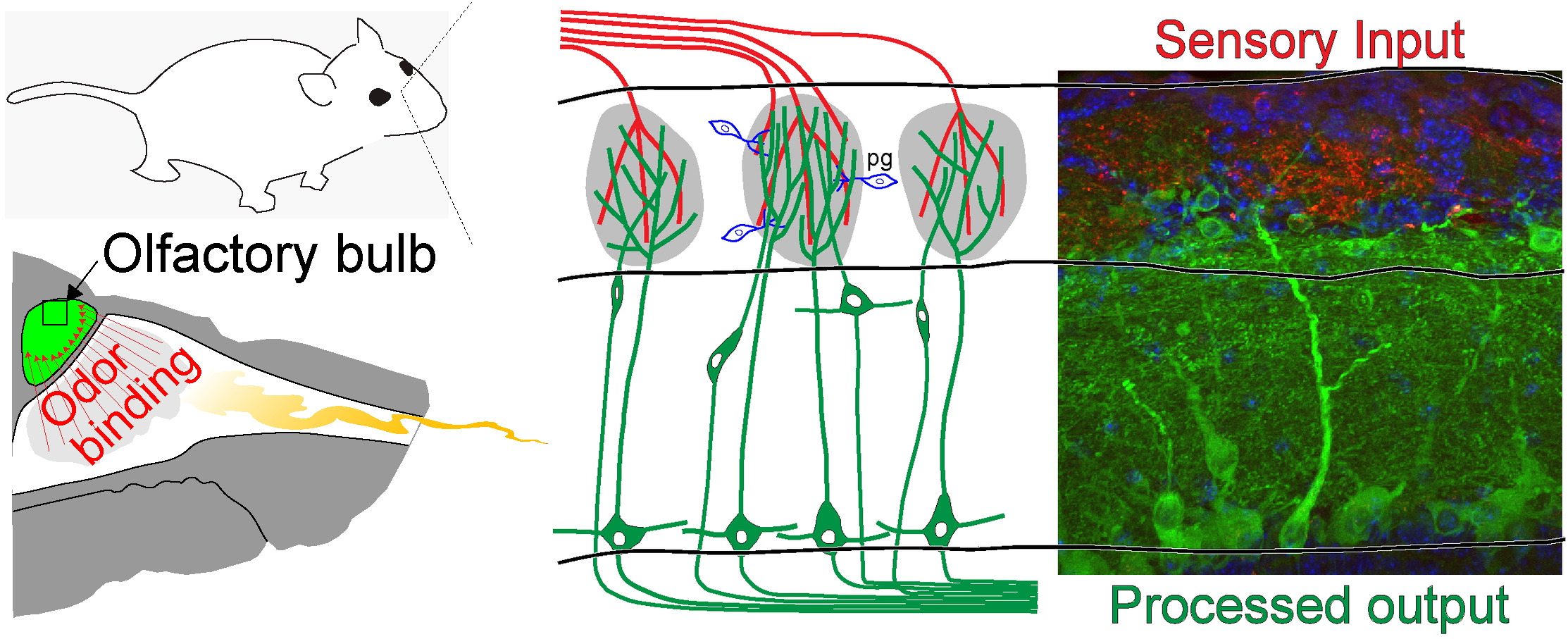
Odor Transduction
Inhaled odors bind to receptors located in the nasal cavity.
Odor Processing
Odor information is sent to the bulb (red) where it is transformed and transmitted to the rest of the brain (green).
We study how sensory information is transformed across this circuit.
Genetically encoded sensors are fluorescent proteins that report cellular activity.
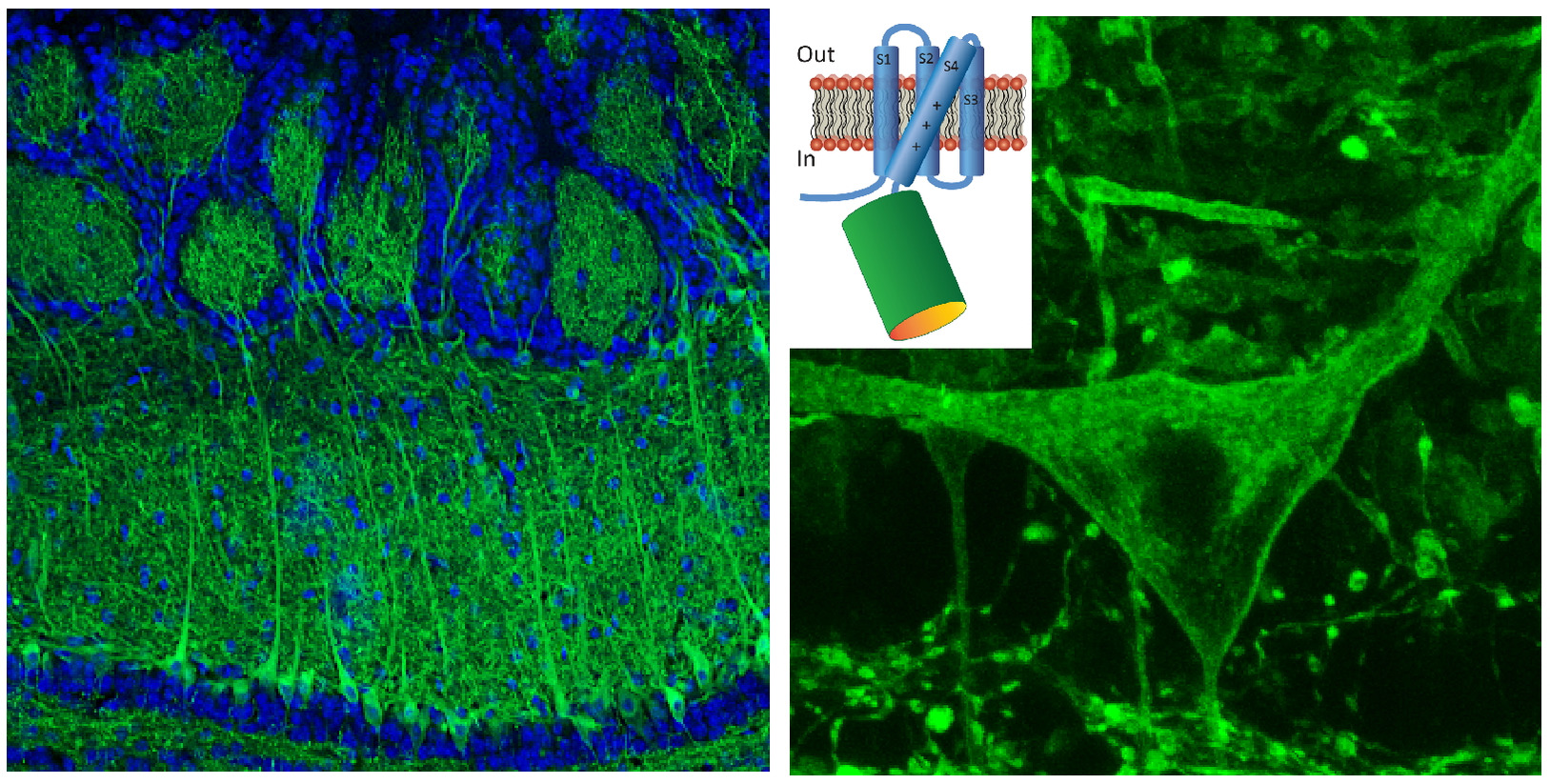
Genetic targeting allows measurements of functional activity from different cell types.
Multi-color 2-photon imaging to simultaneously measure activitiy in different cell types
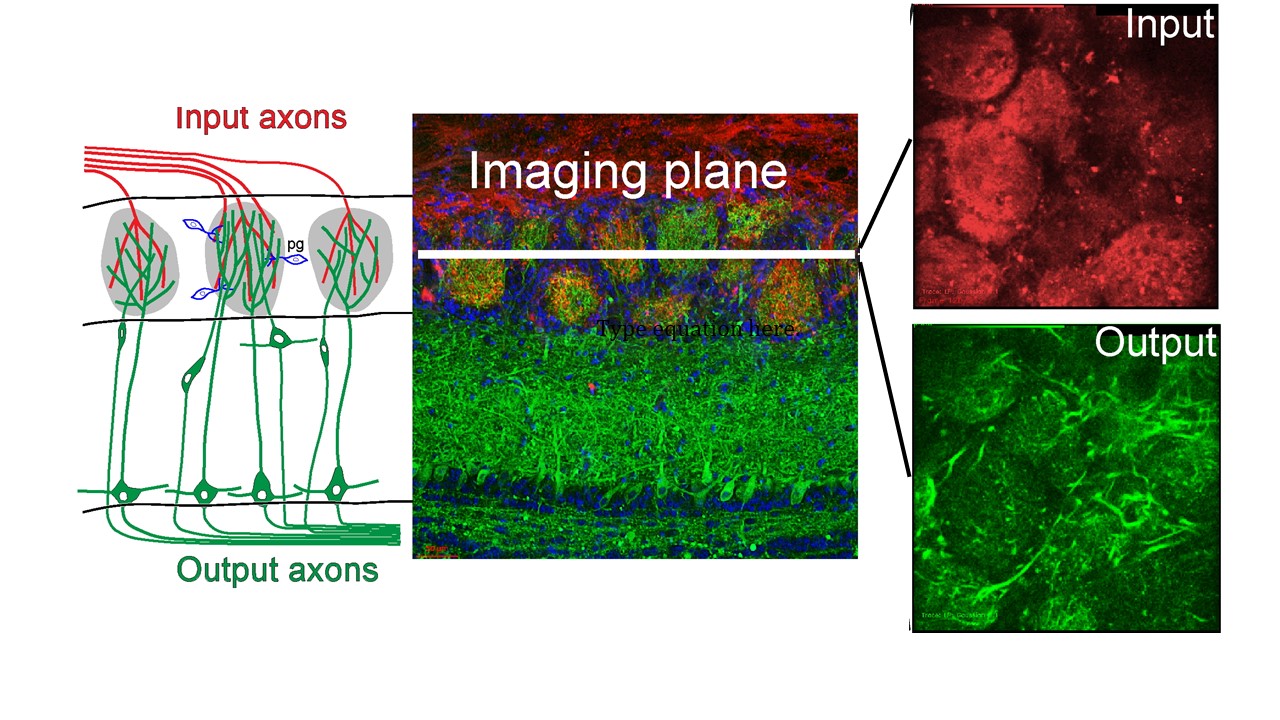
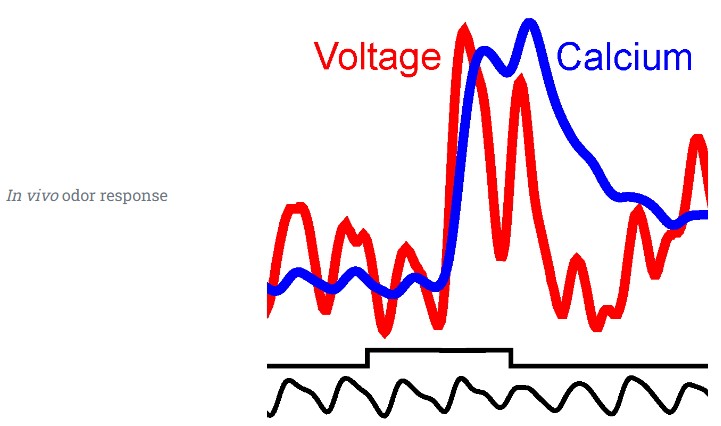
Functional transformations within the bulb
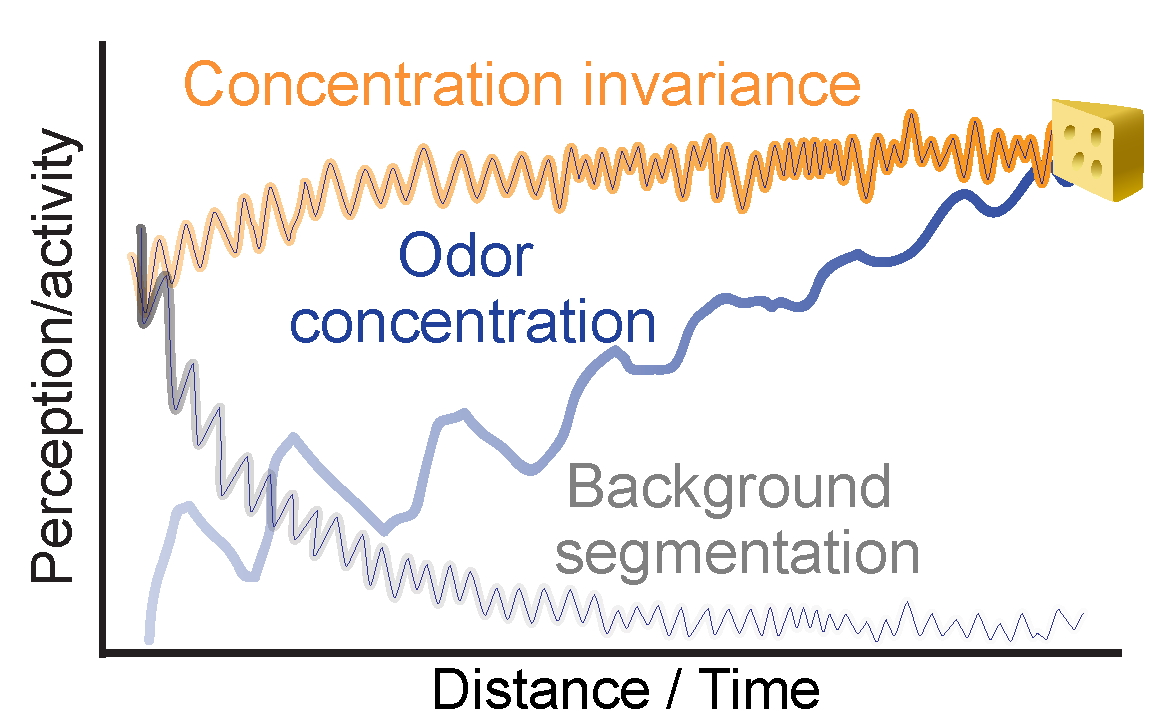
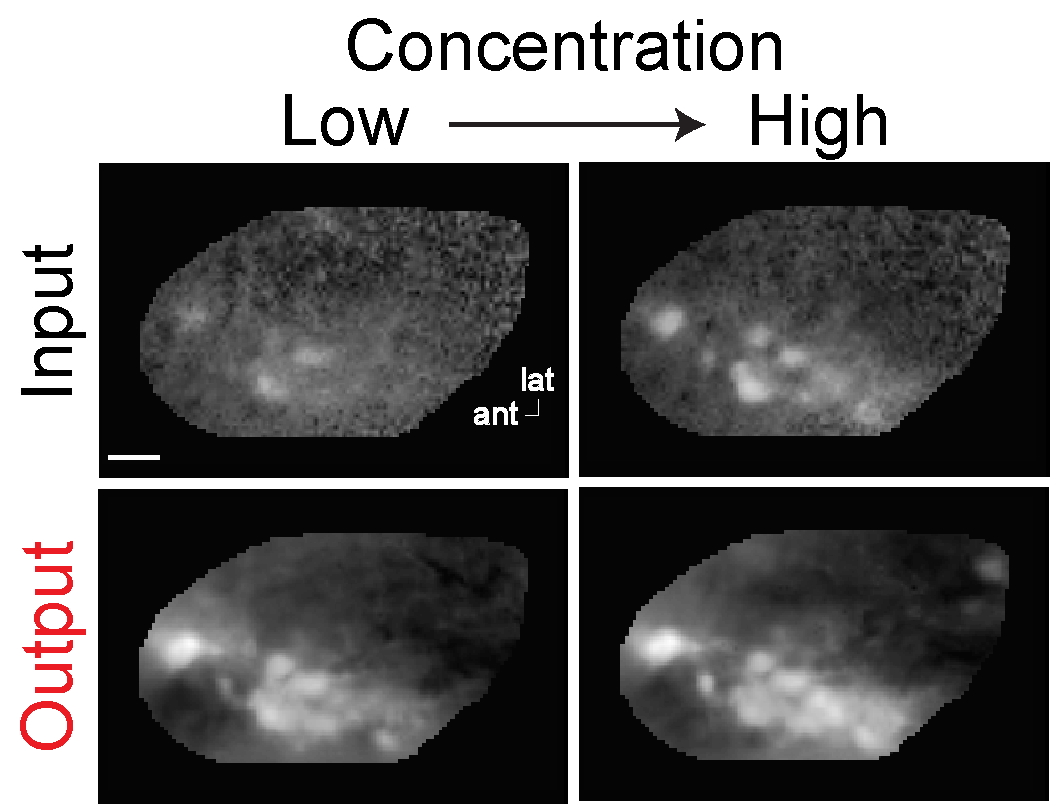
Concentration sensitivity
Odor representations are stable across concentrations.
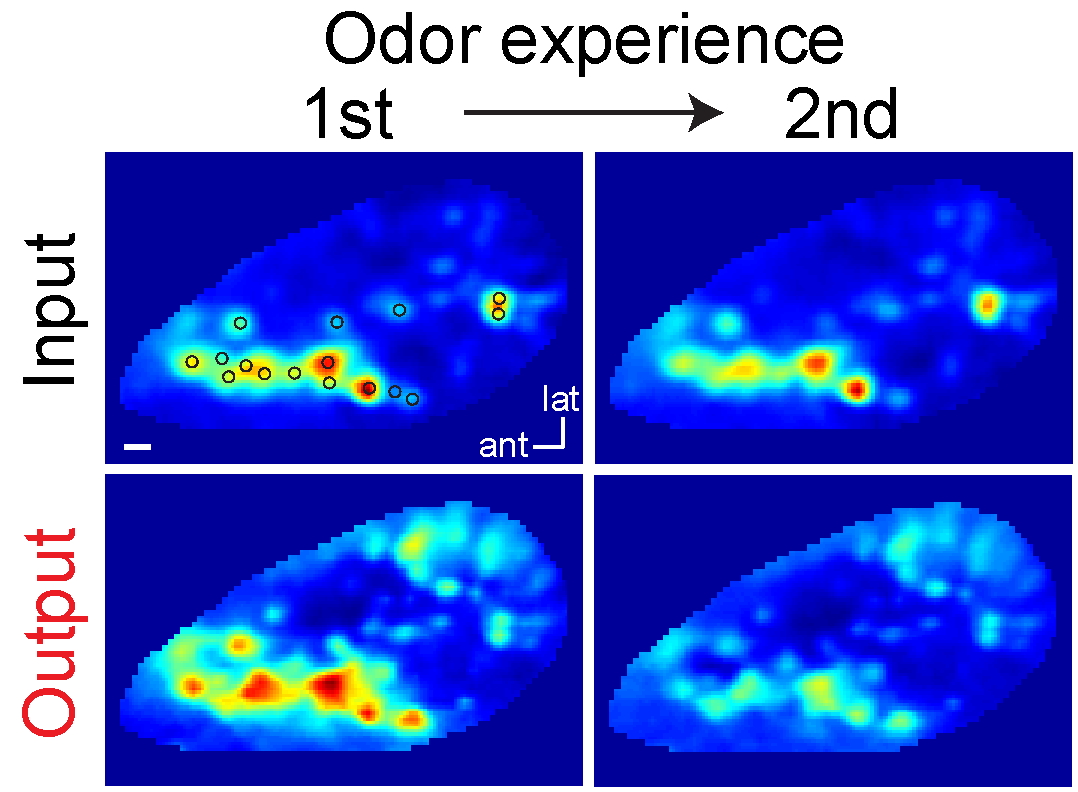
Background adjustments
Bulb processing generates background adjustments.








